SCIENCE SHOOT. . . . .
Even plants like a sugar rush. Or so researchers have discovered while studying the sweetness of sap, which carries sugars from a plant's leaves to other locales, such as the roots. Plants face a tricky balancing act as they load sugar into their sap. Sap with too much sugar is too thick to flow easily, but sap with too little sugar makes for inefficient transport. In today's Journal of the Royal Society Interface, researchers say they devised a mathematical model that predicts the sweet spot for sap: 23.5% sugar by weight. That's far sweeter than Coke, which is 10% sugar. The researchers dug through the literature and found sap sugar-levels for 41 plants, which together average roughly 18%—not far from the model's predictions. The outliers include maize (41%; pictured above) and potato (50%), suggesting that humans have domesticated plants that are on a natural sugar high.
With up to a few dozen holes pockmarking each of its leaves, the Swiss cheese plant (genus Monstera, pictured) is aptly named. But why would these plants, which dwell in the shadows of rainforests in the Americas, limit the leaf area they need to suck up sunshine? New computer simulations reveal that missing pieces may help the plants capture light more dependably in unpredictable situations. In the tropical understory, flecks of sunlight make it through the canopy intermittently and erratically. The holes allow leaves to spread out over greater areas without needing to expend energy and nutrients growing extra leaf area to fill this space. That, in turn, may improve the plant's chances of catching sunflecks, scientists will report in the February issue of The American Naturalist. Future experiments can test this model using grids of light sensors with holes in them to see if they actually can catch as many sunflecks as grids without holes.
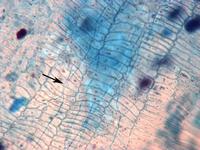
Fungi (blue lines) living among the cells of a grass cause that plant to make more seeds and less pollen. Credit: Jennifer Rudgers Life demands tradeoffs, and plants are no exception. Virginia wildrye, common on U.S. prairies and rangelands, often plays host to a fungus that helps this grass grow. But the plant pays a price. Researchers have discovered that infected plants produce less pollen than their noninfected counterparts. Instead, the fungus causes the rye grass to make extra seeds, which transmit the fungus to the next generation and new locations. This is the first time a fungus has proven capable of manipulating plant reproduction. The finding highlights the complexity of the relationship between hosts and their guests.
Relationships abound in which a plant or animal partners with another organism. That organism, called a symbiont, gets protection from the environment and, in return, sometimes offers some benefit. In the case of Virginia wildrye, the microscopic fungus thrives between the cells of the stem and leaves. It makes a toxin that deters insects and other animals from eating the plant and also protects its host against disease and drought.
But Jennifer Rudgers, a botanist at the University of New Mexico, Albuquerque, who led the new study, began to wonder if there was more to the relationship. Even though Virginia wildrye is hermaphroditic, producing seeds (the female contribution) and pollen (male contribution), the fungus gets passed on to the next generation only in seeds. Pollen is too small to carry the fungus, she notes. "Basic theory predicts that whenever a symbiont is preferentially transmitted through one sex, it will evolve [ways] to overproduce that sex," explains John Werren, an evolutionary geneticist at the University of Rochester in New York who was not involved with the work. The same thing might hold true for an organism's sex cells, such as pollen and eggs. Many insects, for example, host microbes that are "inherited" through the eggs but not through sperm. The microbes often cause more female insects to be produced. They may convert genetic males into females, allow females to produce only daughters without the need for mating, or kill male offspring of infected females so that the daughters have more food. "These 'strategies' enhance transmission of the microbe," Werren says.
Rudgers decided to test whether the fungus was skewing the grass's reproductive efforts in a similar way. She compared pollen and seed production in plants that were infected with the fungus and plants that were not.
Relationships abound in which a plant or animal partners with another organism. That organism, called a symbiont, gets protection from the environment and, in return, sometimes offers some benefit. In the case of Virginia wildrye, the microscopic fungus thrives between the cells of the stem and leaves. It makes a toxin that deters insects and other animals from eating the plant and also protects its host against disease and drought.
But Jennifer Rudgers, a botanist at the University of New Mexico, Albuquerque, who led the new study, began to wonder if there was more to the relationship. Even though Virginia wildrye is hermaphroditic, producing seeds (the female contribution) and pollen (male contribution), the fungus gets passed on to the next generation only in seeds. Pollen is too small to carry the fungus, she notes. "Basic theory predicts that whenever a symbiont is preferentially transmitted through one sex, it will evolve [ways] to overproduce that sex," explains John Werren, an evolutionary geneticist at the University of Rochester in New York who was not involved with the work. The same thing might hold true for an organism's sex cells, such as pollen and eggs. Many insects, for example, host microbes that are "inherited" through the eggs but not through sperm. The microbes often cause more female insects to be produced. They may convert genetic males into females, allow females to produce only daughters without the need for mating, or kill male offspring of infected females so that the daughters have more food. "These 'strategies' enhance transmission of the microbe," Werren says.
Rudgers decided to test whether the fungus was skewing the grass's reproductive efforts in a similar way. She compared pollen and seed production in plants that were infected with the fungus and plants that were not.
When an insect blunders into the sticky tentacles of the carnivorous sundew plant, the leaf curls up into a kind of outer stomach in which the plant digests its catch. This is no mere reflex but a complex chemical system of capturing and devouring live prey, a new study suggests. Working with the South African cape sundew (Drosera capensis, pictured), researchers analyzed the chemicals produced by plants given a meal of live fruit flies. As the leaf engulfed the fly, the plant produced compounds called jasmonates that increased along with the degree of curl, the team reports today in the Proceedings of the Royal Society B. The leaves also curled up when the investigators touched them with a liquid preparation of jasmonate. But small stones or gentle strokes with a brush had no effect, indicating that the curling response is not just a reaction to contact or movement. Nor did dead flies evoke any response (inset). However, crushed dead flies caused the leaves to curl up—suggesting that chemicals released by trapped prey may trigger the production of jasmonates, which turn the leaf into a stomach. Many plants produce jasmonates in defense against munching insects. But that wasn't the case here; fruit flies, which eat rotting fruits and vegetables, have soft mouth parts that don't damage a sturdy, living plant. The researchers suspect that the sundew has adapted a system meant to defend against predators into one that allows the plant to become a predator itself.